The term, “organic chemistry,” is derived from the observation that the chemistry of life / the chemistry of organs inside living things has a certain pattern. That is it involves molecules constructed of atoms of carbon, hydrogen and oxygen. Carbon atoms form the structural framework by linking together in carbon chains. Atoms of hydrogen and oxygen are attached in places stabilizing the molecule and providing important reactivity sites.
There are other elements associated with biochemistry. One that is particularly interesting is nitrogen.
Before I proceed, I want to disclose that I am not specially trained in physical chemistry. The statements that follow are my interpretations from reading information about the chemical properties of nitrogen.
So, nitrogen. We find it in important biological molecules such as proteins, DNA, cell membranes, hormones, and specialty molecules like chlorophyll and hemoglobin. We find it in caffeine and LSD. For whatever reasons, living things continue to employ atoms of nitrogen as part of their ongoing operations.
The good news is that Earth’s atmosphere is rich in molecular nitrogen (N2). Earth’s atmosphere is 78% N2. The bad news is that N2 is very nonreactive. This means that despite its abundance, nitrogen atoms bound together as N2 cannot be engaged in simple organic chemical reactions. Plants, animals, fungi and most bacteria cannot withdraw N2 from the atmosphere and inject it into their internal biochemical reactions.
Why is N2 so resistant to chemical reactions? It turns out that when two atoms of nitrogen are attached to each other as a molecule of nitrogen (N2), the attachment is very, very strong. This is because the atoms are held very tightly by three strong bonds. Like this N≡N. The bonding action occurs very close to the nuclei of these atoms which makes them particularly strong. So, breaking this bond requires a great deal of energy and sophisticated technique. Almost all living things cannot budge this bond.
It turns out that there are some kinds of bacteria that can break the N≡N bond. They are generally described as the “nitrogen-fixing” bacteria, and the cyanobacteria. They possess a nitrogenase enzyme that assists in breaking this bond. They not only break the N≡N bond, but they also attach three hydrogen atoms two each liberated nitrogen atom. This makes the compound known as ammonia (NH3). Ammonia is extremely reactive, and in this form, it is instantly useful as a carrier of reactive nitrogen atoms.
What makes nitrogen atoms so useful in biochemistry? Remember those three bonds that held two nitrogen atoms together? It means that a single nitrogen atom has three easily connected bonding sites. It means that for NH3, it’s easy to swap out a hydrogen atom and insert a carbon. Like in the left-had side of an amino acid, as shown in the image below.
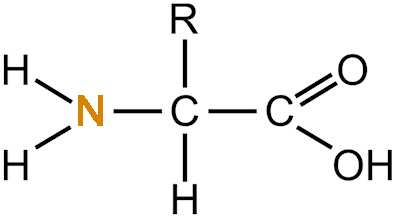
Amino acid
Those now-available strong bonding sites give nitrogen atoms unique characteristics in organic chemistry. And nitrogen atoms give molecules special reactivity characteristics. (I’m sorry. That’s the best I can do.)
Let’s take a look at some examples.
We saw the nitrogen atom in the amino acid above. That nitrogen atom has two hydrogen atoms attached to it on the left, and it is connected to a carbon atom on the right. Well, if we bring in another amino acid on the left, then it is easy to connect the two by connecting to the nitrogen atom. This is called a “peptide” bond. The result is a dipeptide – a simple kind of protein molecule.
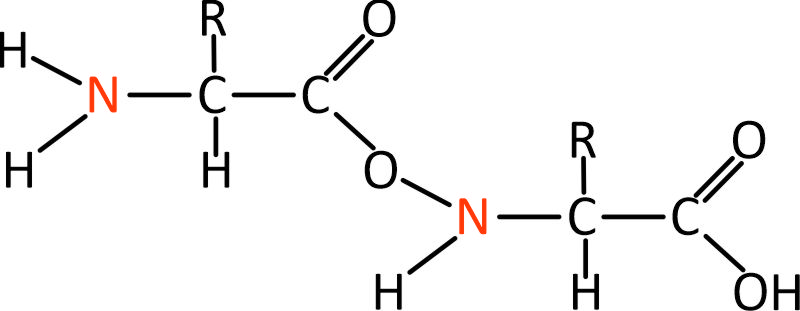
Simple dipeptide protein
Nitrogen atoms are so abundant in the molecules that form the rungs of DNA, that those molecules are referred to as “nitrogenous bases.” See the image below.
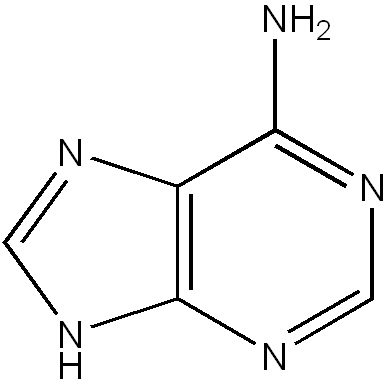
Nitrogenous base.
Chlorophyll (below) is a molecule in plants that captures sunlight in the first step of photosynthesis. Nitrogen atoms form a kind of ring suspending an atom of magnesium in the center. What this means, chemically, I’m not sure.
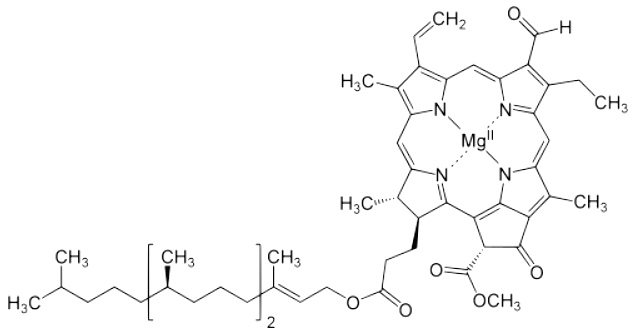
Chlorophyll
One more. Hemoglobin (below). Similar to chlorophyll, the heme group consists of four nitrogen atoms in a ring. At the center rests an iron atom.
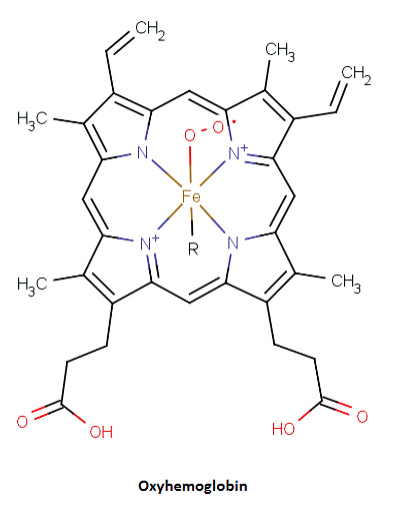
Hemoglobin.
As I said, I’m not trained in physical chemistry, so I can’t wow you with informed discussions on valence states, bond-dissociation enthalpy and oxidation numbers. But in general, nitrogen provides life’s chemistry with versatility and useful chemical reactivity.
Question. If nitrogen-fixing bacteria and cyanobacteria have been converting N2 into ammonia for hundreds of millions of years, how is it possible that we still have so much nitrogen in the atmosphere?
Here is an excellent link that describes the versatility and usefulness of nitrogen.